Tell us your story
Please enter the following information.
We care about your privacy. By clicking the button you accept our disclaimer and privacy policy.
Please enter the following information.
We care about your privacy. By clicking the button you accept our disclaimer and privacy policy.
Last update: December 15, 2017
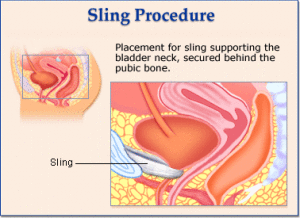
One of the more recent lawsuits that have come up in the world of medical treatment are the Bladder Sling lawsuits. A bladder sling is a medical device that is used to treat stress urinary incontinence, or SUI, in women. A bladder sling is comprised of strips of surgical mesh and is typically inserted through one incision in the vagina and two small incisions in the abdomen.
Stress urinary incontinence is a disorder that primarily occurs in women. This disorder is characterized by the involuntary loss of urine due to physical activities such as sneezing or laughing. Medical experts state that this is because the urethra, which is the tube that carries urine out of the body, is much shorter in women- at about two inches than it is in men- at about ten inches. Additionally, pregnancy and childbirth, and even aging can contribute to stress urinary incontinence.
When the procedure is successful, a bladder sling can actually entirely change the life of a patient. However, in many cases, there are many painful complications that result from this procedure that cause a patient to regret their choice to use this surgery to correct their stress urinary incontinence, and as a result, many people have filed bladder sling lawsuits.
To make a bladder sling, the surgeon uses strips of mesh or other synthetic tissue to create a hammock around the bladder and urethra to keep them closed during normal activities.
In many cases, bladder sling surgery is confused with those surgeries that are performed to correct pelvic organ prolapse, which is a very similar condition that can be repaired with transvaginal mesh. However, even though these surgeries do involve many of the same organs, are very different.
There are a few types of bladder slings that have gained approval by the United States Food and Drug Administration. Some of the most common slings include the following:
This procedure uses a mesh tape which acts as a sling placed under the urethra and held in place by the patient’s tissues instead of being stitched into place. This procedure came out in 1996.
This tension-free procedure has less of a risk of bowel and/or bladder injury when compared to the TVT sling due to the fact that it is not necessary to blindly pass a large needle through the retropubic space when the tape is inserted. This procedure was invented in France, and in 2002, brought to the United States.
This procedure has the lowest risk of complications when compared to the two above because there is no need for groin or abdominal incisions. In this procedure, a very small single incision is made in the vagina and the mesh tape is placed into a u-shape in mid-urethra. This is the newest procedure and began to be used in 2006.
One study done on the bladder sling procedures took 75 women with an average age of 47 years old. The study revealed a complication rate of 21.5 percent. Complications of these procedures included bladder injuries, excessive bleeding, and vaginal extrusion. Basically, the study revealed that TVT seems to be better than TOT one year after surgery. However, TOT seems to be much safer than TVT.
In some cases, serious problems result after bladder sling surgery. Patients reporting issues post-surgery state that they have difficulty urinating for a prolonged period of time or even new symptoms of incontinence- more specifically urgency. The other risks associated with bladder sling surgery include infections at the staple or screw points, injury to organs, or internal bleeding.
The most serious of the complications that result are reported from synthetic slings eroding into nearby organs in the pelvic cavity including the rectum and vagina. Additionally, organ perforation has been reported- this occurs when the bladder sling punctures an organ. Finally, the patient may also experience inflammation, which is an indication that the body rejects the sling.
Another serious complication is mesh contraction, which leads to severe pelvic pain and even painful sexual intercourse.
After having bladder sling surgery, patients should be aware that they will be expected to make some significant alterations to their lives during the period of recovery. They should know that they will not be able to urinate on their own for several weeks and will be required to use a catheter. They will also experience pain, which can be controlled with prescription medications from their physicians. It is recommended that they use OTC fiber supplements for constipation.
They will not be able to perform any heavy lifting for one to three months following the procedure. In order to minimize the risk of infection, they will be required to give up sexual intercourse, taking baths- they can take showers, swimming, or using the hot tub until the stitches completely dissolve, which can take up to two months.

First of all, its safety record has been linked back to a product that was recalled in 1999 by the Food and Drug Administration- Boston Scientific Protegen Sling.
Secondly, ObTape was approved due to its similarity to other tapes used in bladder slings. However, it was actually made of much more dense materials that caused complications. The thicker weave of this product didn’t allow the surrounding tissues to receive the necessary nutrients, which meant that the body couldn’t accept the device easily.
The ObTape was approved by the Food and Drug Administration under the 510 (k) process. This means that there was no pre-market testing because of its similarities to other products already on the market.
Mentor has now sold its surgical urology business to Coloplast, which was later acquired by Johnson and Johnson.
There have been hundreds of bladder sling lawsuits filed since 2008 due to the defects of Mentor’s ObTape. These bladder sling lawsuits have been combined into a MDL or Multi-District Litigation in the US District Court for the Middle District of Georgia. This MDL is being presided over by Judge Clay D. Land and allows the pre-trial proceedings to be consolidated into one. It is slowly moving forward, and after the first trial got underway, Johnson and Johnson began settling with plaintiffs.
Around 35,000 women in the US have had a sling implant using ObTape, however- only a fraction of these have filed bladder sling lawsuits as a result.
We’ve seen various settlements in 2015 and 2016, for example with American Medical Systems. The settlement amount in these cases was typically around $40,000, but seriously injured women have received amounts of $250,000 or more.
Bloomberg reported in January 2016 that Johnson & Johnson agreed to pay more than $120 million to resolve 2,000 to 3,000 lawsuits. To give you an idea, this would come to an average of $40,000 to $60,000 per case.
Page Contents
Drugwatcher helps you to stand up against producers of dangerous drugs and medical devices.